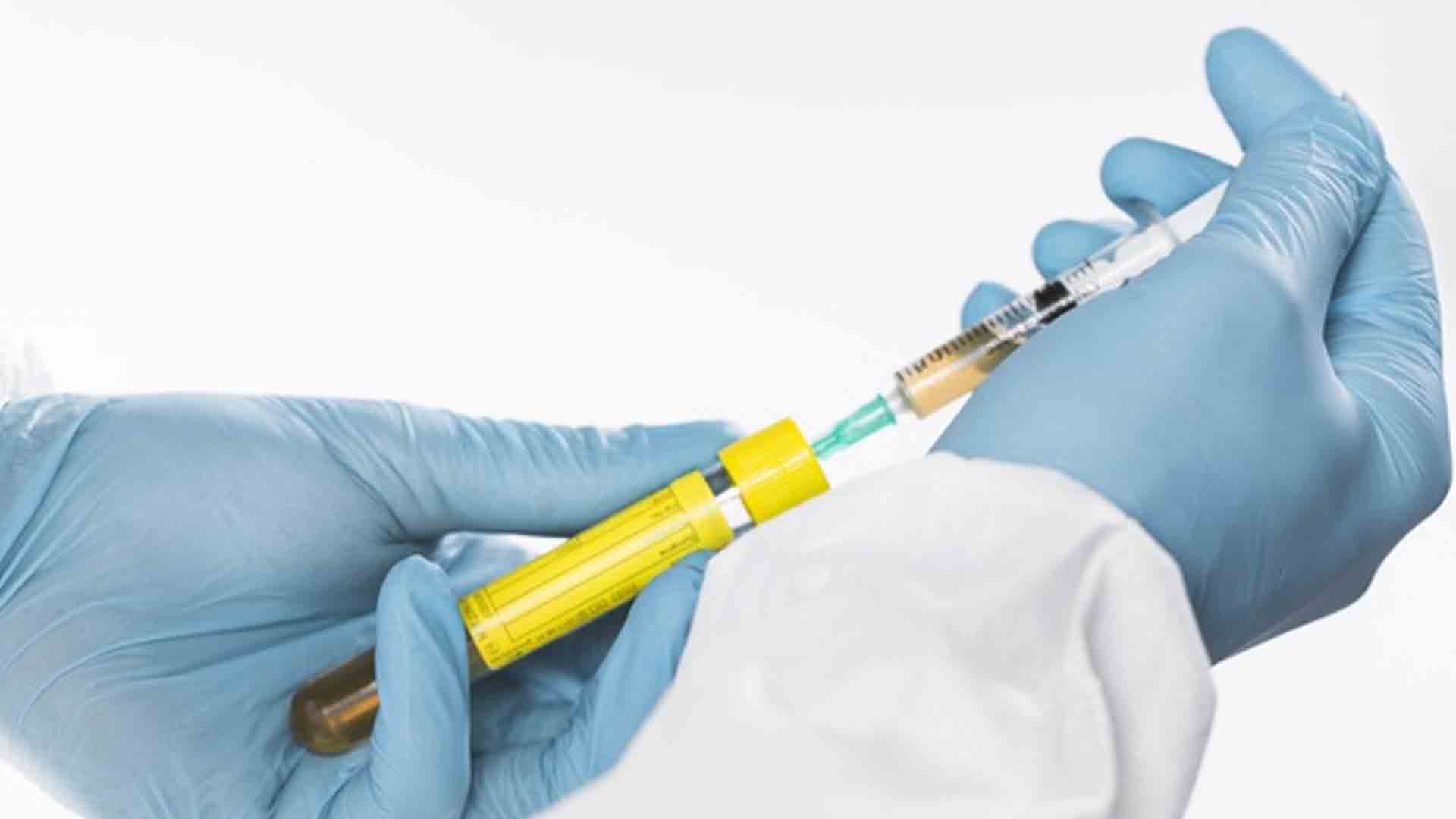
Safeguards such as surveillance mechanisms and pilot implementations will be in place to ensure the safety of coronavirus disease 2019 (Covid-19) vaccines even in the absence of Phase 4 clinical trials, a health official said Monday.
In a virtual press briefing, Health Undersecretary Maria Rosario Vergeire said the Department of Health “is part of the group which has recommended for the exemption for the Phase 4 clinical trial”.
“This is for us to expedite the process so we can use the vaccines as soon as possible once it reaches the country, (and), this accelerated process has been done already in various parts of the world for ebola and meningitis vaccines,” Vergeire said.
To expedite the procurement of vaccines, a waiver allowing the government to purchase Covid-19 vaccines even if they have not undergone Phase 4 clinical trials was included by lawmakers in the proposed Bayanihan to Recover as One Act.
The fourth phase or “post-marketing surveillance trials” delve on the evaluation of a drug’s or vaccine’s long-term safety and effectiveness.
Vergeire said the Health Technology Council recommended that the safeguards must include surveillance mechanisms monitoring for effectiveness, safety, and quality according to World Health Organization recommendations.
“Secondly, there should be a list of all who will be given the vaccine. We’ll set up a database for those who will receive the vaccine and it will include their primary details like age, demographic characteristics, and contact information,” she said.
Citing that the vaccines are new, Vergeire noted that there must also be informed consent from those who will use the vaccine and community engagement.
“Magkakaroon ng pilot areas kung saan uumpisahan ang pagbibigay ng bakuna at saka natin palalawigin ang mga pilot areas na ito (There will be pilot areas where [we’ll] start the administration of the vaccine and then we’ll expand these pilot areas),” she added. (PNA)