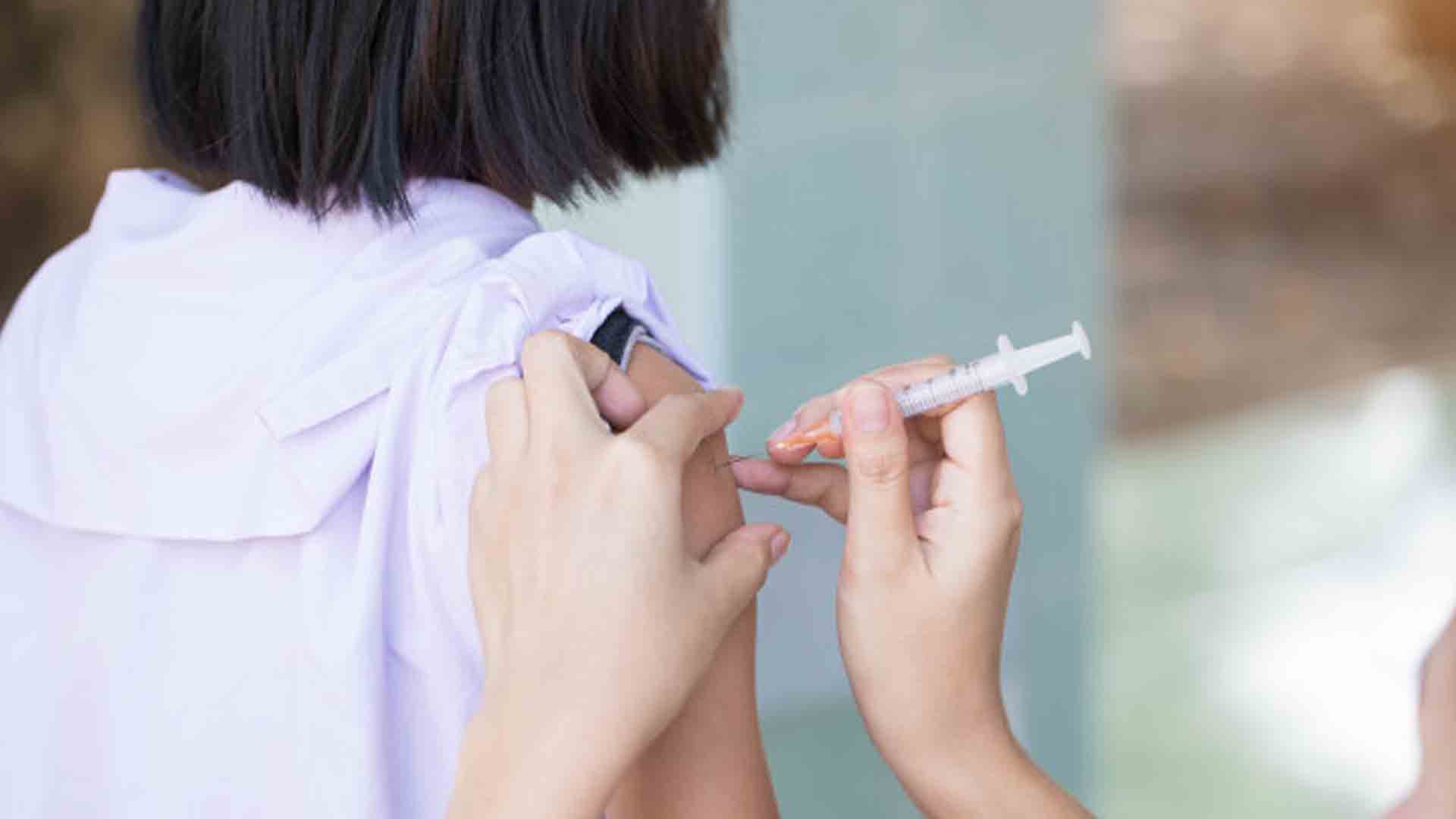
The government will prioritize the use of Pfizer and Moderna brands of Covid-19 vaccines for the country’s pediatric vaccination program, National Task Force (NTF) against Covid-19 chief implementer, Secretary Carlito Galvez Jr., said Thursday.
“We have decided na yung other vaccine lang like yung Moderna and Pfizer iko-concentrate namin yun sa tinatawag nating (We have decided that the other vaccines such as Moderna and Pfizer will be concentrated to the) pediatric vaccination, considering that it will also be used as boosters for 12 to 17 years old,” Galvez, also the vaccine czar, said at the sidelines of the ceremonial rollout of the government’s “Resbakuna sa Botika” program in Pasig City.
Galvez said the government expects the increase of vaccine demand for pediatric vaccination as they will soon start vaccinating children aged 5 to 11 years old.
“We really have constraints with other [brand] of vaccines, considering that the other vaccines can only be used for primary series,” Galvez said. “We only have two vaccines that can be used for 12 to 17 years old while only one vaccine brand for 5 to 11 years old.”
The government is also waiting for the emergency use authorization (EUA) of Covid-19 jab for zero to four years old.
“As of this moment nakikita namin na tataas ang requirement ng (we see an increasing requirement for) pediatric vaccination because starting already the 5-11 and later pagkanagkaroon na ng EUA ang 0-4, we will start it again so nakikita natin malaki ang requirement natin (we see that we have a greater requirement),” Galvez said.
The government is mulling to acquire more than 60 million formulated or lower-dosed Covid-19 jabs to be administered, as primary series and booster shots, to a much younger population.
The country, Galvez said, will be needing 12 million doses for 12 to 17 years old age bracket, over 15.56 million for 5 to 11 years old as well as 11.11 million doses for infants and toddlers or 0-4 population.
Galvez earlier said some 780,000 lower-dosed Pfizer vaccines for the youngest age group so far are expected to arrive by end of January, followed by 1,632,000 doses in February.
He added Sinovac Biotech Ltd.’s EUA application for 3 to 17-year-olds now underway. (PNA)