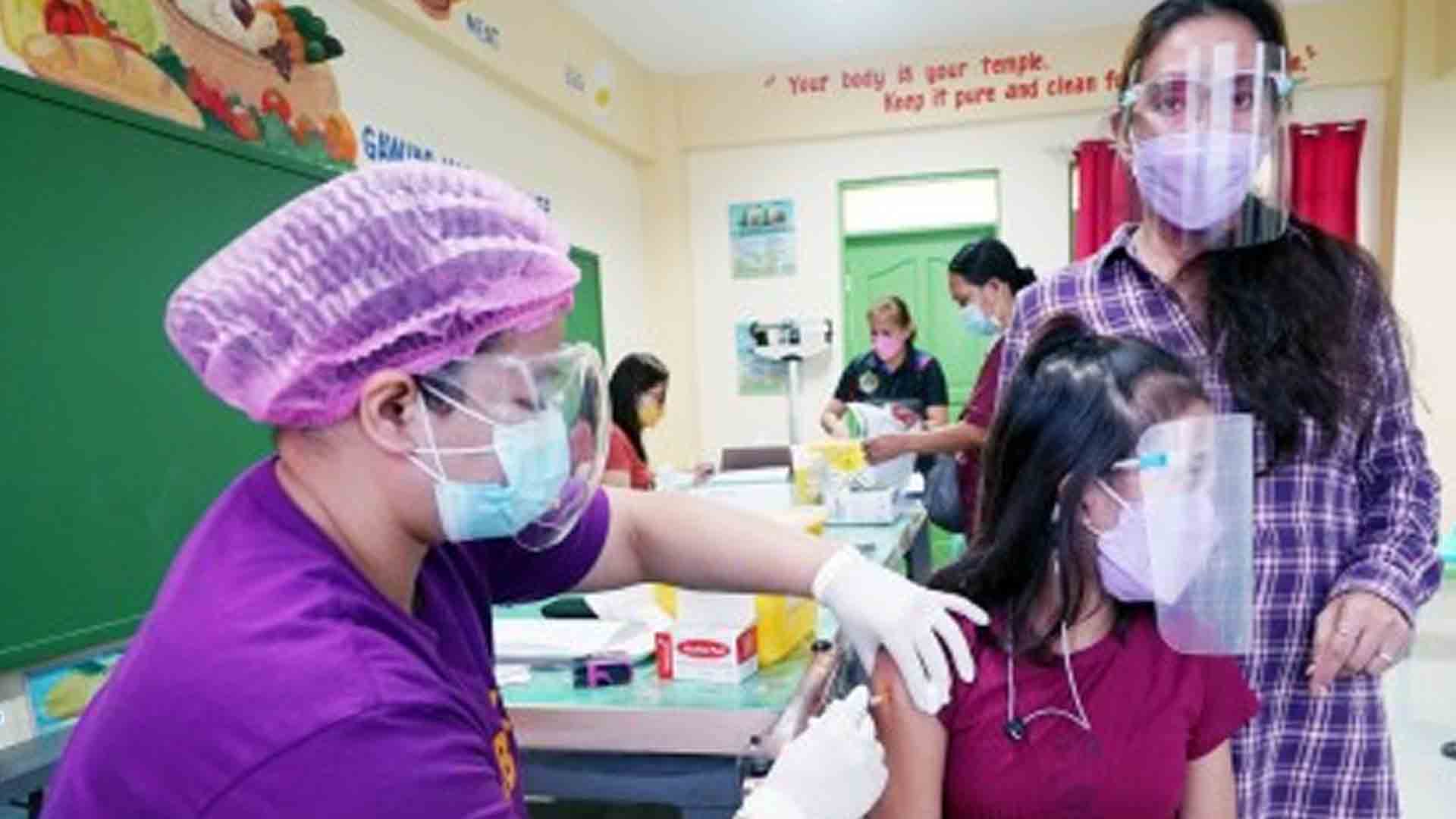
The government is coordinating with other drug manufacturers to expand the emergency use authorization (EUA) of coronavirus disease 2019 (Covid-19) vaccines for pediatric vaccination.
National Task Force (NTF) Against Covid-19 chief implementer, Secretary, Carlito Galvez Jr., said they are eyeing the use of other brands of coronavirus jabs for the younger population.
“Kami po ay nakipag-ugnayan na po sa ibang mga brand para po magkaroon po tayo ng EUA dito po sa ages 3 to 11 (We are coordinating with other brand manufacturers so that we can have EUA for ages 3 to 11 years old),” Galvez reported to President Rodrigo Duterte during the prerecorded Talk to the People aired on state-run People’s Television Network late Monday night.
The Food and Drug Administration has already approved the use of Pfizer-BioNTech and Moderna vaccines for the 12 to 17 age bracket.
Galvez said the government targets to start the administration of primary doses to some 12.5 million minors aged 5 to 11 years old within the second quarter this year.
The lower dosages of the Pfizer vaccine for the age bracket “might be available by the end of January,” according to Galvez.
About 11.11 million aged zero to four years old may also be inoculated by April while some 10.2 million children in the 12 to 17 age group should have already received booster doses by June. (PNA)